Design Specifications
- Cost: To be competitive with existing public screening methods and still be accessible to regional healthcare facilities in India, we intend to price it at $100 (approximately Rs. 6000) or below. This price point allows the device to be competitive with individual monitors and still present a significant price decrease compared to currently employed public automated monitors.
- Ease of use: This specification addresses a combination of factors including convenience and the ability to use the device without prior training. Our device should be usable by literate individuals with no medical training. The results should be displayed to the user automatically without the need for interpretation on the patient’s part. To enhance the convenience of the device for healthcare workers, it should also be able to store unique readings from at least 100 patients for future use or integration into health records.
- Accuracy: Readings should fall within 0.67 kPa (5 mmHg) of the measurement provided by a calibrated automated monitor. The primary function of our device is to serve primarily as a screening tool, identifying individuals that should seek further medical attention and consultation. Therefore, the device should be able to provide reasonable distinction between different categories of normo- or hypertension. Because we intend to for this device to be used for public screening purposes, an automated oscillometric monitor, the most commonly used public screening method seen in the US, was chosen as the benchmark for accuracy. The accuracy bounds were chosen based on the British Hypertension Society protocol for assessment of new devices.
- Rapid: To increase convenience and facilitate evaluation of several subjects in a reasonable timeframe, our device should be able to record blood pressure within 20 seconds of being set up. This target was based on the speed of the most common public automated monitor on the market, the LC300 from Life Clinic, which takes approximately 1 minute to return results.
- Portability: The device will be designed with use outside of a clinical setting in mind. Therefore, it should be easy to transport it to whatever location the operator deems appropriate for public screening. To meet this goal, the entire system should weigh no more than 2.27 kg (5 lb).
- Reusability: The device and its components will be reusable to conserve resources in low-income areas.
Sources:
1. Ogedegbe, Gbenga, and Thomas Pickering. "Principles and Techniques of Blood Pressure Measurement." Cardiology Clinics 28.4 (2010): 571-86. Web.
2. "Sentry Health Monitors LC 300." Sentry Health Monitors LC 300. N.p., n.d. Web. 23 Oct. 2014.
3. Ancillare. "BpTRU - BPM-100 Clinical Blood Pressure Monitors." BpTRU - BPM-100 Clinical Blood Pressure Monitors. Ancillare, 2011. Web. 19 Sept. 2014. <http://ancillare.com/bptru/products.html#bpm_100>.
Design Overview
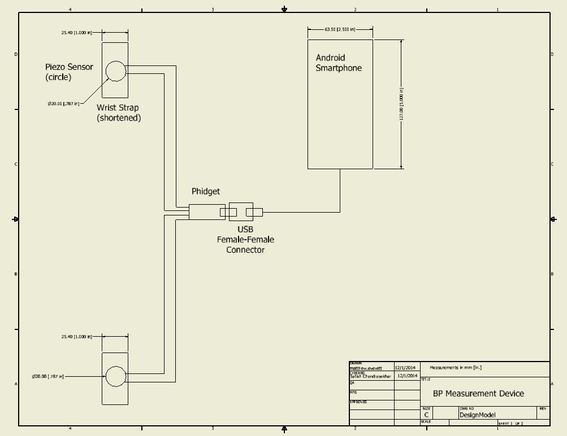 Device design
Device design
The device will acquire pulse data from two locations on the patient's body (wrist and forearm) using piezoelectric sensors. This analog data will then be transferred to an Android smartphone through the use of an analog-digital converter and USB. The Android phone will then filter and use this data to calculate pulse-wave velocity, which can be converted into an approximate value of blood pressure, which will be displayed to the patient using an application on the Android device.
Design Components
The components for the current device prototype include:
Use of these components meets all design specifications, including cost, speed, weight, and ease of use.
- 2 x muRata Piezoelectric Sensors (Model Number 7BB-20-6L0)
- 2 x Velcro Straps (Zhongshan Liyao Weaving Company Model Number 55033)
- 1 x Phidget Interface Kit 2/2/2 (Model Number 1011_0)
- 1 x USB Female-Female Connector (Monoprice Model Number 362)
- 1 x Android Smartphone (Jivi JSP 20)
Use of these components meets all design specifications, including cost, speed, weight, and ease of use.
Design Safety and Risk Analysis (DesignSafe Report)
| group38designsafereport.xls | |
| File Size: | 24 kb |
| File Type: | xls |
